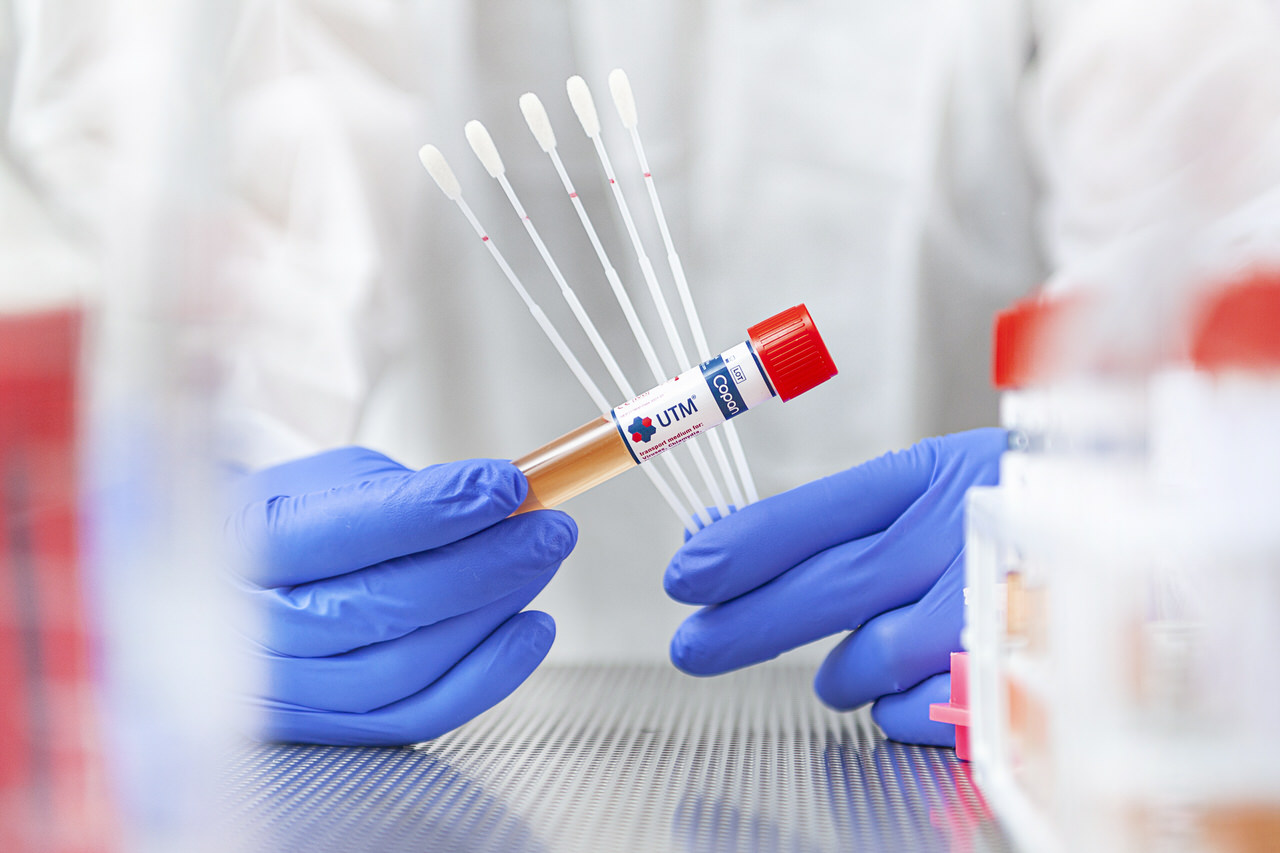
PodSwab™ is the 6 ml UTM® tube paired with five nasal flocked swabs that allows for pooling up to five different individual samples in one single tube.
PODSwab ™ combines all the strengths of UTM® medium in a practical kit designed to ease the collection and processing of samples, especially in low-incidence diseases. By pooling samples, laboratories can significantly increase the number of individuals tested while using the same amount of resources and at a lower cost.

The concept behind PodSwab™ is simple. If the pooled sample tests negative, all the pooled patients are considered negative – by performing one test instead of five separate tests. Conversely, if the pooled sample is positive, each person must be individually retested to identify who was positive.
UTM® PodSwab™ is compatible with downstream antigen detection and molecular-based assays, and can be processed manually or with automated laboratory platforms such as Copan Universe™. Noteworthy, the combination of PodSwab™ with UniVerse™ pooling operational mode opens up the opportunity to automatically pool and test together up to 20 samples.


UTM® PodSwab™ quintuples the efficiency of one of our most opulr products, UTM®

Ensure a quick, capillarity-driven sample uptake and a superior elution of the biological specimen, expanding downstream diagnostic testing capabilities.

PodSwab™ increases the number of individuals that can be tested using the same amount of resources, increasing the testing capacity of every lab while saving time and reducing the cost per single test.

UTM® allows to preserve the viability of viruses, Chlamydia, Ureaplasma, and Mycoplasma for 48 hours at both controlled room and refrigerated temperature; it can also be stored at -80°C for one year.

Many scientific studies have demonstrated pooled testing to be an efficient methodology for screening in the case of low-incidence phases of COVID-19.
Check here the summary of UTM® PodSwab™ features and contact us to discover the product code that best suits your need-mix and available in your Country.






Write us to satisfy your curiosity, get information or start a great collaboration!
Use our search tool and be surprised by how easy it is to get right to the point.