FecalSwab™ is intended to collect fecal specimens and preserve enteric pathogenic bacteria’s viability during transport to the testing laboratory. In the laboratory, FecalSwab™ samples are processed using standard clinical laboratory operating procedures for culture.
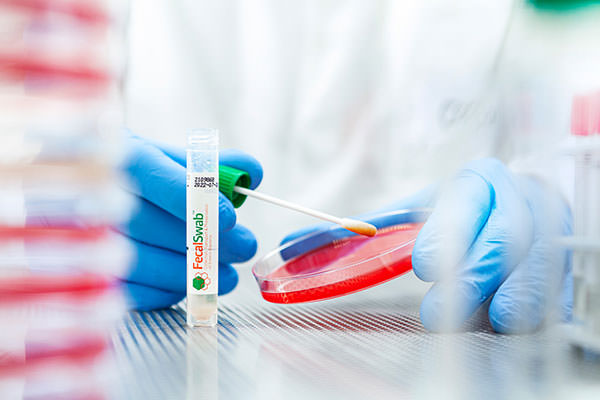
FecalSwab™ is a modified Cary-Blair medium specifically designed for the recovery, preservation and detection of enteric pathogens, available with two collection devices: a standard flocked swab and a specialized one with stopper for the collection of anal, perianal and feces.
In the laboratory, FecalSwab™ collected samples are compatible with Copan WASP® automated processing and suitable enteric bacteria culture.

FecalSwab™ is a convenient system for transporting fecal samples!

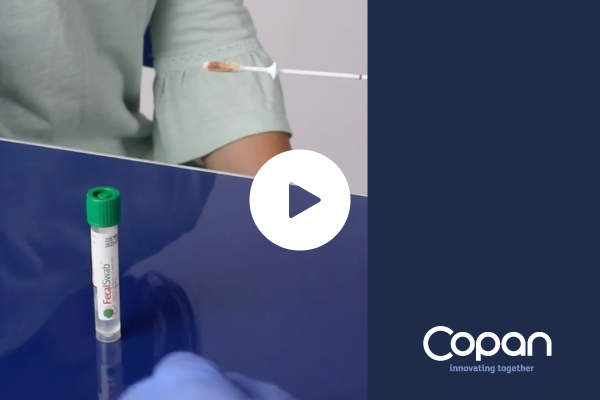
Fecalswab™ can be used by medical staff to transfer a small quantity of sample from the primary stool collection container, or to directly collect a rectal swab sample.
FecalSwab™ simplifies fecal sample collection, transport, and processing. Its shatterproof tubes are a compact and neat alternative to large, bulky transport containers.

FecalSwab™ is compliant with CLSI M40-A2 Quality Control
for Microbiological Transport System standards.

FecalSwab™ preserves collected specimens for 48h at room temperature or 72h at refrigerated temperature. In the case of C. difficile culture investigation, Copan FecalSwab™ preserves collected specimens up to 24h at room temperature
and 48h at refrigerated temperature.

The VI-PAK metallic foil and plastic film barrier block oxygen entry, preventing unwanted oxidation of the transport medium.
The combination of FecalSwab™ with FLOQSwabs® expands your possibilities even further by ensuring an unmatched specimen collection in many anatomical collection sites. Discover why we call them “the perfect collection device.”

This product line groups a variety of product codes, each optimized to address specific needs.
Check here the summary of these features and contact us to discover the product code that best suits your need-mix and available in your Country.
Always faithful to our scientific approach, we are eager to hear the proof of the success of our efforts and commitment.



Collection, transport, and preservation device for enteric pathogens

FecalSwab™ is intended to collect fecal specimens and preserve enteric pathogenic bacteria’s viability during transport to the testing laboratory. In the laboratory, FecalSwab™ samples are processed using standard clinical laboratory operating procedures for culture.
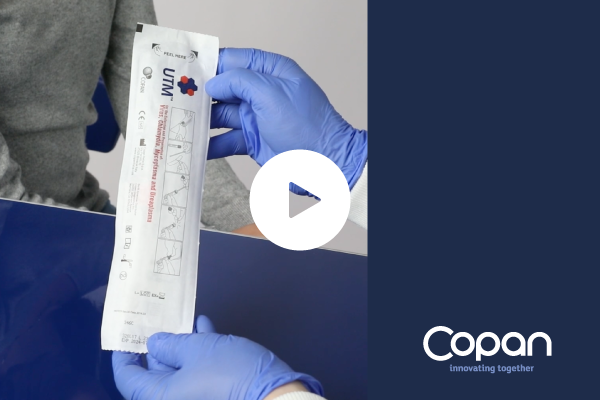
Copan Universal Transport Medium® (UTM®) system is intended for the collection, transport, and preservation of clinical specimens containing Viruses, Chlamydia, Mycoplasma, and Ureaplasma.

Write us to satisfy your curiosity, get information or start a great collaboration!
Use our search tool and be surprised by how easy it is to get right to the point.